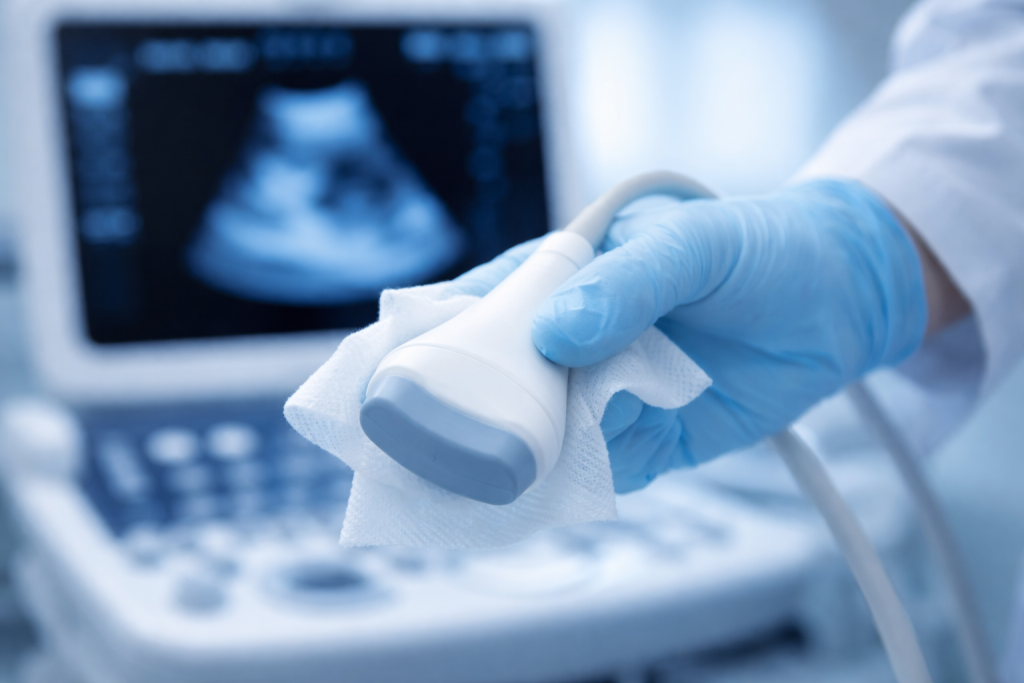
Healthcare-Associated Infections (HAIs) are among the most preventable — and costly — challenges facing modern hospitals. Among the many potential vectors of transmission, ultrasound transducers represent a significant and frequently underestimated risk. A single probe used across multiple patients without the correct barrier and decontamination protocol can transmit bacterial, viral, and fungal pathogens. Nulatex medical-grade probe covers are specifically designed to eliminate this risk.
This guide outlines the evidence-based best practices for ultrasound hygiene, aligned with infection control standards.
Understanding the Risk: Semi-Critical vs. Non-Critical Devices
In infection control, medical devices are classified by their level of patient contact, following the Spaulding Classification system:
- Non-Critical Devices: Contact only intact skin (e.g., external abdominal probes). These require low-level disinfection between uses.
- Semi-Critical Devices: Contact mucous membranes or non-intact skin (e.g., transvaginal, transrectal, or intraoperative probes). These require high-level disinfection AND a probe cover for every single use.
The distinction is crucial. Using a substandard probe cover on a semi-critical probe is a significant infection control failure, regardless of whether the probe is visually clean.
The Three-Step Protocol for Endocavity Probes
International guidelines recommend the following sequence for every endocavity examination. For a detailed visual walkthrough, see our step-by-step probe cover use and disposal guide.
- Cover Before Use: Always apply a medical-grade probe cover before insertion. Inspect the cover for visible defects, tears, or pinholes. Avoid using a standard condom as a substitute — reservoir tips trap air which can compromise the scan.
- Remove and Inspect After Use: Remove the probe cover carefully to avoid contaminating the probe body. Inspect the used cover for any tears or fluid breach. If a breach is found, the probe must be escalated to full high-level disinfection before its next use.
- Clean and Disinfect the Probe: Even with a probe cover, the transducer must be wiped down with an approved low-level disinfectant wipe after every procedure and subjected to high-level disinfection at regular intervals or after any probe cover failure.
Choosing the Right Probe Cover: Key Specifications
Not all probe covers are equal. For a comprehensive breakdown of materials and sizing, read our guide: Probe Covers — A Closer Look at Materials, Sizes, and Applications. When sourcing covers for your facility, verify the following:
- Sterility: For endocavity use, choose individually sealed, single-use probe covers
- Material: Medical-grade latex or latex-free alternatives for facilities managing latex-sensitive patients
- Flat-end design: Essential for acoustic integrity — eliminates the air pocket created by reservoir-tip alternatives
- Uniform wall thickness: Prevents image distortion caused by uneven barriers
- Compliance: Confirm the product meets ISO 13485 medical device manufacturing standards
Staff Training: The Human Factor in Infection Prevention
Even the best probe cover cannot compensate for poor technique. Clinical facilities should ensure that all ultrasound practitioners receive regular, documented training on:
- Correct donning and removal of probe covers to prevent contamination
- Recognition of product failure and correct escalation procedures
- Proper disinfectant contact times — a wipe placed on a surface for two seconds does not achieve the kill time required on the product label
- Documentation of any suspected cover breach for HAI tracking purposes
Nulatex medical-grade probe covers are manufactured to ISO 13485 standards and are available in sterile dispenser boxes and also bulk non-sterile packs to suit every clinical workflow. Delivery is available across Malaysia. Browse our full ultrasound probe cover resource library for more clinical guidance.